Why Your Stress Hormones Don't Reset Overnight When You Haven't Slept Enough
Apr 20, 2026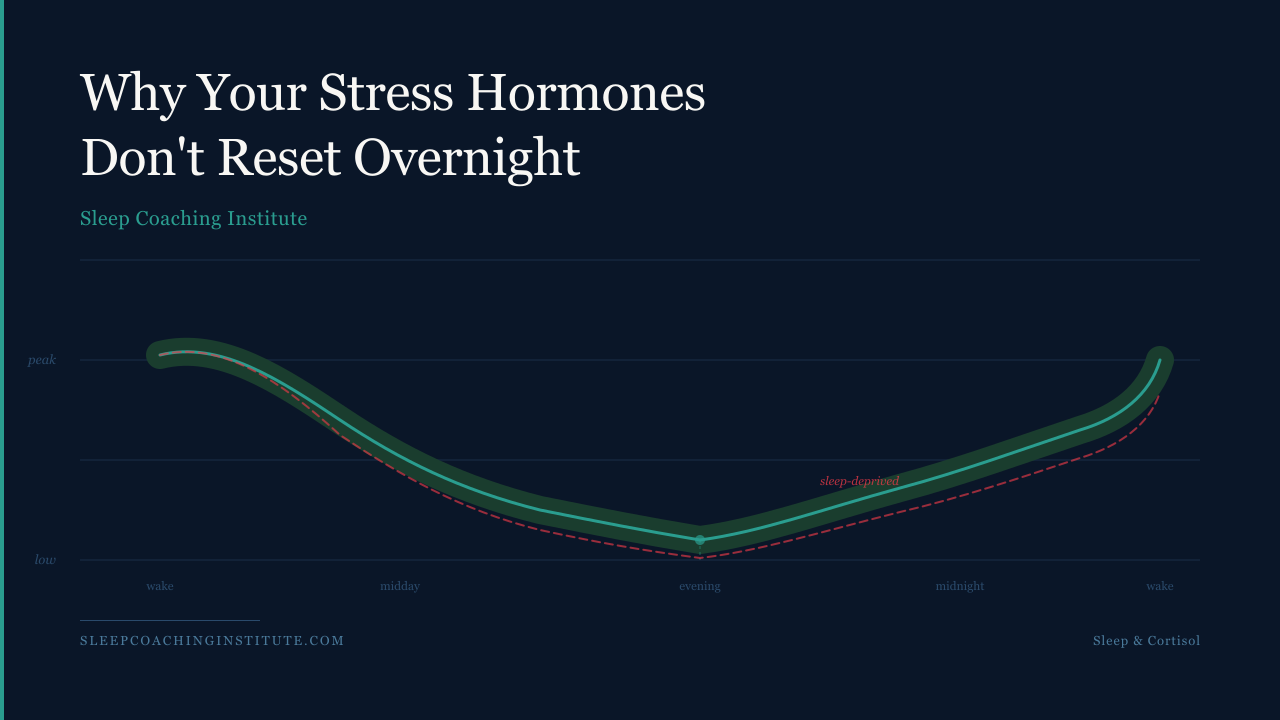
Key Findings
Partial sleep deprivation raises evening cortisol levels by 37 to 45 percent the following day, delaying the onset of cortisol's nightly quiet period by at least one hour. (Leproult et al., 1997)
After six nights of four-hour sleep, the rate of evening cortisol decline slows approximately sixfold compared to fully rested subjects, a pattern typically associated with older age and insulin resistance. (Spiegel et al., 1999)
Sleep-deprived individuals show both higher resting cortisol and an amplified cortisol response to acute stressors, suggesting the HPA axis is structurally sensitized by insufficient sleep. (Minkel et al., 2014)
Most people assume stress causes poor sleep. That part is true. What most people do not realize is that the relationship runs just as hard in the other direction. Cut your sleep short enough, for long enough, and your body begins producing stress hormones as if something is wrong, even when nothing is. You wake up already braced for a threat that does not exist.
The system responsible for this is called the hypothalamic-pituitary-adrenal axis, or HPA axis. Think of it as a three-way relay between your brain and the adrenal glands that sit above your kidneys. Under normal conditions, sleep acts as a brake on this relay. Deep sleep specifically quiets the system, suppresses cortisol output, and gives the axis a chance to reset for the next day. When sleep is cut short, that brake releases too early. The relay stays active longer than it should. Cortisol, which should be falling through the evening, keeps circulating instead. And the longer this pattern repeats, the more sensitized the system becomes, reacting harder to the same inputs. A University of Chicago research team spent years documenting exactly how this unfolds, and the findings are worth understanding in detail (Balbo et al., 2010).
Cortisol itself follows a natural daily arc, much like the arc of daylight. It peaks near waking, high and purposeful, then tapers steadily through the afternoon and reaches its lowest point in the early evening before beginning a slow overnight rise that primes you for the next morning. That rhythm is not merely convenient biology. It is the body's primary mechanism for regulating inflammation, blood sugar, immune function, and cognitive alertness across a 24-hour period. When sleep is cut short, that arc does not simply flatten. It breaks.
Research out of the University of Chicago documented this failure with unusual precision. When subjects experienced either partial or total sleep deprivation, their plasma cortisol levels during the 6 to 11 p.m. window were significantly higher the following day, rising 37 percent after partial deprivation and 45 percent after total deprivation compared to baseline. The onset of cortisol's natural quiescent period was delayed by at least an hour (Leproult et al., 1997). That delay matters more than the numbers suggest. The evening drop in cortisol is the body's signal that recovery can begin. Push that signal back, and you compress the recovery window before the next sleep cycle starts. It is like trying to cool a room when someone keeps propping the window open.
The problem compounds with repetition. A landmark study published in The Lancet followed healthy young men through six consecutive nights of four-hour sleep. By the end of that period, the rate at which cortisol declined in the late afternoon and evening had slowed by approximately sixfold relative to fully rested conditions. The authors noted that this pattern was characteristic of much older adults, and that elevated evening cortisol levels were likely to promote insulin resistance, a metabolic risk factor with downstream implications for obesity and type 2 diabetes. When the same subjects were given recovery sleep, these abnormalities resolved (Spiegel et al., 1999). Six nights. That is not a long timeline.
What this tells us is that sleep debt does not accumulate passively, the way a stack of unopened mail piles up on a counter. It actively recalibrates how the stress hormone system operates. The setpoint shifts. The system gets louder. And because the body is efficient, it gets better at the pattern it keeps practicing, even when that pattern is working against you.
The sensitization effect is equally important to understand. When sleep-deprived participants were exposed to an acute social stressor, their cortisol response was not just elevated at baseline. It was amplified. Compared to rested controls, sleep-deprived subjects showed both higher resting cortisol and a stronger reactive cortisol spike to the same stressor. The HPA axis had not merely drifted upward. It had become more reactive, responding harder to the same provocation (Minkel et al., 2014). This means poor sleep does not just make you feel more stressed. It physiologically increases the hormonal response to events that would otherwise be manageable. The dial has been turned up, and ordinary friction starts to feel like a real threat.
The relationship also runs the other way, and this is where the cycle becomes genuinely difficult to interrupt. Elevated cortisol suppresses slow-wave sleep, the deep stage that would otherwise be doing the work of calming the HPA axis. Sleep loss raises cortisol. Elevated cortisol degrades sleep quality. Each night with poor sleep primes the next one to be harder. Understanding this loop is one of the most useful things you can do, whether you are working with clients or trying to address your own sleep, because it explains why willpower alone rarely fixes the problem (Balbo et al., 2010).
For anyone curious about the broader picture of how sleep shapes recovery, hormone regulation, and chronic disease risk, the Sleep Coaching Institute's resources for individuals provide an accessible entry point. The physiological connections between sleep and metabolic health are also covered in depth within SCI's professional certification curriculum, because these mechanisms sit at the center of clinical sleep dysfunction in client populations.
There is one additional layer worth naming. The circadian rhythm of cortisol is orchestrated by a small cluster of neurons in the hypothalamus called the suprachiasmatic nucleus, the brain's master clock. Think of it as the conductor keeping every section of the orchestra on the same beat. Sleep timing, light exposure, and consistent wake behavior all feed cues into that conductor. When those cues become inconsistent, the beat drifts. Chronic circadian misalignment, the kind that comes with irregular sleep schedules, shift work, or persistent late bedtimes, produces sustained disruptions to the cortisol rhythm that go beyond what a single bad night creates. The phase and amplitude of the cortisol curve both shift in ways that extend risk well beyond the morning after (Wright et al., 2015). This is not a problem that resolves on weekends.
Understanding these mechanisms clarifies why poor sleep is not just a symptom of stress. It is one of its causes, and one of its most direct physiological amplifiers. That distinction matters differently depending on whether you are working with clients or working on your own sleep, which is why both perspectives deserve their own treatment below.
For Professionals
What This Means for Health and Wellness Professionals
The HPA axis disruption described above is not an edge case. It is the baseline physiology of a substantial portion of the clients likely sitting in front of you. Adults who report feeling wired at night, struggling to wind down after low-stakes evening activities, or waking between 2 and 4 a.m. with racing thoughts are frequently describing a cortisol profile that has shifted rightward. The evening drop has been delayed, and the quiescent period has been compressed.
From a clinical standpoint, this shifts how you assess the stress-sleep relationship. Rather than treating stress as a cause and sleep difficulty as the downstream effect, the evidence supports treating them as bidirectional. A client who says they cannot sleep because they are stressed may equally be stressed because their sleep has eroded HPA regulation. Intervening only on stress management without addressing sleep architecture directly will often produce limited results.
The sensitization finding carries direct implications for client education. When a sleep-deprived client describes feeling disproportionately reactive to minor frustrations or perceiving their daily load as heavier than it used to feel, they are likely describing a real neuroendocrine shift rather than a coping deficit. Naming that mechanism clearly, explaining that their system has been dialed up rather than that they are failing to cope, can reduce self-blame and meaningfully increase engagement with sleep-focused interventions.
Sleep posture and environment also intersect here. Fragmented sleep from positional discomfort reduces slow-wave sleep time, which is the specific stage most associated with HPA suppression. Even clients who spend adequate time in bed may be accumulating cortisol dysregulation if their sleep quality is compromised by pain, poor support, or a non-conducive environment.
For professionals who want to build formal competency in the clinical assessment of sleep-stress interactions, the Certified Sleep Coach (CSC) program covers HPA axis physiology, sleep architecture, and client-facing protocols across five modules with 0.6 continuing education units approved by NASM and AFAA.
For Individuals
How to Use This for Your Own Sleep
If you recognize yourself in any of this, you are not alone. Most people who feel anxious in the evenings, lie awake despite being exhausted, or wake in the early hours of the morning are experiencing some version of cortisol rhythm dysregulation. The biology described above is not abstract. It is the experience of a nervous system that never fully received the hormonal signal that the day is over. Your body is still waiting for the all-clear that poor sleep keeps delaying.
The most direct thing you can do is protect the early and mid-evening hours from inputs that keep cortisol elevated. Think of your evening as a runway, not a hard stop. The body needs gradual deceleration, not a sudden switch from full speed to sleep. Physical exercise later than two to three hours before bed, emotionally intense conversations, bright overhead lighting, and screen-based content that produces urgency or arousal are all capable of sustaining cortisol elevation into the hours when your body needs it to fall. Dr. Allyson La Barbara, a Stanford-trained board-certified physician specializing in neurodevelopment and nervous system regulation, puts it directly: “Cognitive or emotional stimulation late in the day may override physical tiredness.”
That override effect is real and it is physiological, not a character flaw. Feeling tired but unable to sleep is often a sign that cortisol has not yet cleared sufficiently to allow the sleep initiation process to proceed. You are not broken. The timing is off.
Consistency in wake time is one of the most powerful tools you have for stabilizing the HPA axis rhythm. Your cortisol morning peak is partly anchored to your habitual wake time. Vary that time by more than an hour from day to day and you pull the anchor loose, resulting in a flatter, less predictable curve throughout the day and a less decisive evening drop. Sleeping in on weekends feels like recovery. For cortisol rhythm, it often functions more like jetlag.
Darkness in the bedroom, cooler temperatures, and a decompression buffer of 30 to 60 minutes before sleep all support the cortisol decline your body is working to complete. These are not optional extras. They are the conditions under which the biology actually works.
If you want a structured, evidence-based program to address sleep-wake regulation from the ground up, The CORE System covers circadian reset, sleep drive, nervous system regulation, and environment across four modules with lifetime access, at $199 one time.
Citations
Balbo, M., Leproult, R., & Van Cauter, E. (2010). Impact of sleep and its disturbances on hypothalamo-pituitary-adrenal axis activity. International Journal of Endocrinology, 2010, 759234. https://doi.org/10.1155/2010/759234
Leproult, R., Copinschi, G., Buxton, O., & Van Cauter, E. (1997). Sleep loss results in an elevation of cortisol levels the next evening. Sleep, 20(10), 865–870. https://doi.org/10.1093/sleep/20.10.865
Minkel, J., Moreta, M., Muto, J., Htaik, O., Jones, C., Basner, M., & Dinges, D. (2014). Sleep deprivation potentiates HPA axis stress reactivity in healthy adults. Health Psychology, 33(11), 1430–1434. https://doi.org/10.1037/a0034219
Spiegel, K., Leproult, R., & Van Cauter, E. (1999). Impact of sleep debt on metabolic and endocrine function. The Lancet, 354(9188), 1435–1439. https://doi.org/10.1016/S0140-6736(99)01376-8
Wright, K. P., Jr., Drake, A. L., Frey, D. J., Fleshner, M., Desouza, C. A., Gronfier, C., & Czeisler, C. A. (2015). Influence of sleep deprivation and circadian misalignment on cortisol, inflammatory markers, and cytokine balance. Brain, Behavior, and Immunity, 47, 24–34. https://doi.org/10.1016/j.bbi.2015.01.004
Sleep affects everything. If you found this article useful, you will find even more inside the SCI library, including practical tools for professionals and individuals navigating real sleep challenges.
Stay connected with news and updates!
The science of sleep keeps moving. So do we. Join the SCI newsletter for evidence-based insights, new research, and practical tools delivered directly to the people who need them most.
Your information is never shared. Unsubscribe anytime